Flies Don’t Like REMmy: How Understanding the Sleep Cycle of Flies Benefits our Understanding of Humans

Despite sleep being a biological necessity, humans still have not fully grasped why they need it. Human technology has advanced tremendously over the past few decades, and yet we have only recently begun to understand all of the benefits sleep provides us. However, humans are not the only creatures that require sleep. Every living animal on this planet requires some form of sleep that allows them to repair their cells and conserve their energy. Some of these animals that require sleep are the Drosophila, a genus of flies more commonly referred to as fruit flies. Their sleep shares many similar traits to human sleep, such as similar stages of NREM sleep and brain waves (Beta when awake, Alpha when in light sleep, and Theta when in deeper sleep). They even share similar responses to the same hormones in each brain, such as melatonin-inducing sleep in both creatures. Yet, flies do not share the same REM sleep stage and Delta brain waves as humans, which means that flies cannot enter the same deep sleep that humans undergo nor produce dreams in their sleep. There are many aspects of sleep in humans that we still do not understand, but analyzing the sleep cycle and behavioral patterns in flies can help us better understand the true benefits and reasons behind sleep for humans. In this paper, the Drosophila’s sleep behavior patterns will be assessed to determine the physical effects of sleep on the flies and their correlation to human sleep. Then, I will analyze the genetic traits and mutations to determine the source of sleep disorders and the true causes of sleeping habits. Finally, I will summarize the research on the sleep effects and similarities between humans and flies using brain wave findings.
Drosophila act differently in some aspects from humans in their sleep, such as different physical behaviors and reactions during their sleep and arousal periods. According to a study done by Michaela Joyce and a group of researchers on the circadian cycle and homeostatic regulation of seven different Drosophila species, the species share similar if not identical circadian cycles but completely different homeostatic regulations. The circadian cycle is an internal clock that regulates physical and mental behaviors throughout the day, whereas homeostatic regulation is the bodily function that maintains a certain internal environment in response to the outside environment. Both bodily systems affect one another, but are independent of each other. In this study, they analyzed seven Drosophila species in a series of tests, including the monitoring of the percentage of flies sleeping at different points in the day (Figure 1), the tracking of movement probabilities during each stage of sleep (Figure 2), the observing of the effect of sleep deprivation on their sleep hours (Figure 3), and the observing of the effect of male-to-male interaction on their sleep hours (Figure 3). As shown in the figures below, the sleep percentages and patterns of each of the Drosophila species were recorded to be used as the baseline for comparison, and then the experiment analyzed the rebounding abilities of each species, such as sleep deprivation and male-to-male interactions, to determine the homeostatic regulation capabilities of each species.

Figure 1: The sleep percentages of the seven different Drosophila species based on gender and
daylight hours. (Joyce, M., Falconio, F. A., Blackhurst, L., Prieto-Godino, L., French, A. S., &
Gilestro, G. F.). https://doi.org/10.1038/s41467-024-49501-9
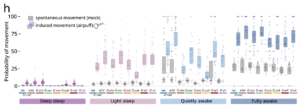
Figure 2: The probability of spontaneous and induced movement in the seven species of
Drosophila based on the sleep stage. (Joyce, M., Falconio, F. A., Blackhurst, L., Prieto-Godino,
L., French, A. S., & Gilestro, G. F.) https://doi.org/10.1038/s41467-024-49501-9

Figure 3: On the left, the asleep percentages were taken for before, during, and after the effect
sleep deprivation had on the hours slept by the seven different Drosophila species. On the right,
the percentages of sleep were taken before, during, and after the effect of male-to-male
How Understanding the Sleep Cycle of Flies Benefits our Understanding of Humans 5
interactions had on the hours slept by the seven different Drosophila species. (Joyce, M.,
Falconio, F. A., Blackhurst, L., Prieto-Godino, L., French, A. S., & Gilestro, G. F.)
https://doi.org/10.1038/s41467-024-49501-9
Each species of Drosophila had a different sleep reaction to sleep deprivation and male-to-male interaction based on how their homeostatic regulations fixed their lack of sleep. The reason homeostatic regulations are different between species of Drosophila is most likely due to the different climates in which each species is located, which dictates how the flies respond to their surrounding environment (i.e., different predators and daylight hours determining how long flies sleep for in a specific given period). This is either caused by the specific evolution of gene pools within the flies that breed or by individual flies reacting in their own way to their surrounding environments. Humans’ homeostatic regulations are impacted by their gene pools and surrounding environments, similar to flies, but they experience homeostatic regulations more in their REM sleep stage through “a propensity or pressure for REM sleep builds up in its absence and triggers a REM period if sufficient pressure has accumulated” (Park & Weber, 2020). Despite humans having homeostatic regulations that affect different parts of the sleep cycle compared to Drosophila, homeostatic regulations remain a crucial part of an animal’s sleep cycle. This begs the question of whether humans and flies are impacted more heavily by their genetics or by their living conditions. In order to determine which has the more dominant effect on the homeostatic regulations of Drosophila, another study was conducted to further explore the genetics of Drosophila and how mutations affect their sleep.
Genetic traits are one of the most important factors in the function of homeostatic regulations. In a recent study, several screenings of Drosophila were done to genetically select Drosophila with sleep gene mutations that impacted their abilities to rebound from sleep deprivation. Sleep deprivation is a shared skill between humans and flies, where if the creature lacks sleep from their previous session, the body will produce a tired feeling that prompts the body to get additional rest in a future sleep session. These gene mutations prohibit the Drosophila body from recovering this way, and it was done by separating the mutated Drosophila from the normal ones to get a sample of only mutated Drosophila. By observing these mutations, the researchers could analyze and compare the shortened sleep gene to the normal sleep gene. They found that the flies had worsened reaction times, impaired cognitive memory, and shortened life spans due to the lack of hours slept (Cirelli & Bushey, 2008). This is due to sleep being heavily affected by the genetic makeup of an individual, which impacts the function of the homeostatic regulations of said individual. If the genetic coding is incorrect, the homeostatic regulations will be hindered in function, which will lead to very negative effects on the creature.
A lack of homeostatic regulations also causes the Drosophila to have worsened arousal periods
(i.e., the length of time it takes for a creature to move from a lower frequency sleep state to an
awake state). Drosophila will have an arousal period that has “decreased with the time it has
rested,” leading to Drosophila having longer arousal periods in sleep deprivation and worsened
chances to escape predators in the wild (Xu et al., 2021). However, the severity of short sleeping
genes are affected by the location of the fly and the surrounding landscape in which they are located.
One could argue that an environment’s physical barriers impact the gene pools of the Drosophila the greatest, making physical climate the more dominating influence on sleep quality. However, the location of the experiments shows that the genetics of an animal affect their sleep abilities more than the circumstances in which an animal is located, as shown by the fact that genetics had a greater impact on the sleep quality of Drosophila and that the conditions in which the Drosophila were living would not have prompted worse sleeping conditions. And due to similar sleep genetic structures between humans and flies, as shown by the genetic screening of Drosophila, it is possible to draw the conclusion that humans share the same impact that flies have from genes. Flies even have a 75% identical genetic match to humans in disease-causing genes, which makes flies a primary candidate to study for sleep diseases (Pfizer). This means that flies share similar genetic sleep disorders to humans, such as insomnia, which is a sleep disorder that makes it difficult for the individual to fall asleep. This causes genetic mutations in flies, including insertional mutagenesis from a transposon inserting a genetic fragment into the gene sequence and changing the transcription of the sequence, and chemical mutagenesis from chemicals such as ethyl methanesulfonate, altering the base structure of the DNA.
Since humans and flies share similar genetic structures, it makes mutations in the gene sequence for sleep testable on flies and applicable to humans. Additionally, since there is a global spread of genetic diversity for humans due to travel, it is easier to see the direct ties genes have to the effects of sleep on humans. This means that in order to get a better understanding of human sleep, the genetic structure of both flies and humans should be examined more thoroughly. Since sleep is heavily tied to brain activity, the next spot that should be examined is the brain and the waves produced during each stage of sleep.
The activity of the human brain shows the benefits and causation of sleep for humans. In a case study done by Melvyn HW Yap and a group of researchers on the fluctuating brain activities of flies, they found that flies share more similar alterations in brain activity to vertebrates than scientists originally thought. Most vertebrates, including humans, have the ability to go through sleep stages while in slumber and change between each stage to fulfill different bodily requirements. Vertebrates have what is called Slow Wave Sleep, where the brain produces waves in the one to four hertz range, i.e., delta waves that theoretically allow for “downscaling synapses or for clearing metabolites from the brain” (Yap et al., 2017). REM sleep, which stands for Rapid Eye Movement, is the opposite of Slow Wave Sleep, where the brain gets to a similar active level as if it were awake and leads to motor skill improvement and dreaming. Sleep in humans involves a fluctuation between slow waves and REM to tend to different bodily needs based on priority. Since Slow Wave Sleep and REM sleep alternate frequently, REM sleep can have properties of high frequency activity and/or low frequency activity based on regions of the brain that are related to dreaming (Siclari et al., 2017). This creates a wave-like pattern of frequency activity when plotted on a line graph with time.
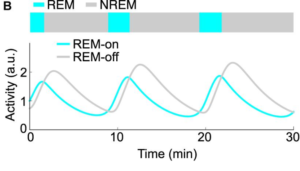
Figure 4: The graph of brain activity based on the sleep stage the brain is in, such as REM or
NREM sleep. (Park, S.-H., & Weber, F.) https://doi.org/10.3389/fpsyg.2020.01662
Figure 4 from a different study done by Sung-Ho Park and Franz Weber shows similar results in the brain activity of humans during sleep, with the constant changing of sleep stages causing a rise and fall in high and low frequencies. Drosophila, due to lacking REM sleep and the rapid eye movement capabilities to dream, do not experience the same kind of slow wave sleeping. However, flies still show patterns of oscillating waves and activity. Using wavelet analysis to determine “how LFP [local field potential] frequencies changed through time,” it was found that the oscillation was only prevalent in completely sleeping flies compared to active flies (Yap et al., 2017). They also found that the oscillations were associated with the transition from one sleep stage to the next, which is likely the same case for humans due to the brain waves constantly changing into the low frequency range (Yap et al., 2017). Therefore, flies and humans share similar brain activities despite the specific waves operating on different frequencies, which further proves that fly sleep is very closely related to human sleep. In analyzing the specific frequency activities, such as the seven to ten hertz range, being tied to the mechanical repair and stimulus, it can be determined that humans share similar frequency activities and functions, and that both flies and humans fluctuate in brain activities to best suit their bodily needs.
Despite the physical differences between flies and humans, the similar homeostatic regulations and patterns between flies and humans in response to sleep deprivation and intraspecies interactions help explain the sleep deprivation humans experience. The homeostatic regulation is also tied to the genes of the individual, which means that the sleep quality of an individual largely depends on the genetic sequence they were given. This means that all living creatures, including flies and humans, are impacted more by genetics rather than the circumstances in which the animals live when it comes to the length and quality of sleep. This explains the sleep disorders that humans experience, being tied to genetics and the different homeostatic regulations found within each individual, since homeostatic regulations are related to gene sets. Additionally, oscillating brain waves and activities during sleep in both humans and flies indicate a shared ability to adjust the body to fix and repair different bodily functions based on the needs of the body. The fluctuation happens in repeated patterns between the brain and sleep stages to cover a wide range of repair functions.
Even with the benefits sleep in flies has given to the understanding of sleep in humans, we still do not know the full functions of sleep. In order to get the full picture, analysis of other creatures and brain functions during each sleep stage is necessary. Another part of sleep that still needs tons of research is dreaming, since we know even less about the reason behind dreams and how it impacts sleep. Future studies pursuing dreams could provide benefits to our understanding of sleep, and maybe even complete the picture and cure sleep disorders. Despite the gaps in our knowledge, we are getting one step closer to understanding ourselves.
References:
Cirelli, C., & Bushey, D. (2008). Sleep and wakefulness in Drosophila melanogaster. Annals of the New York Academy of Sciences. https://doi.org/10.1196/annals.1417.017
Joyce, M., Falconio, F. A., Blackhurst, L., Prieto-Godino, L., French, A. S., & Gilestro, G. F. (2024). Divergent evolution of sleep in Drosophila species. Nature Communications. https://doi.org/10.1038/s41467-024-49501-9
Krause, A. J., Simon, E. B., Mander, B. A., Greer, S. M., Saletin, J. M., Goldstein-piekarski, A., & Walker, M. P. (2017). The sleep-deprived human brain. Nature Reviews.Neuroscience, 18(7), 404-418. https://doi.org/10.1038/nrn.2017.55
Park, S.-H., & Weber, F. (2020). Neural and homeostatic regulation of rem sleep. Frontiers in Psychology, 11, 1662. https://doi.org/10.3389/fpsyg.2020.01662
Pfizer. How Genetically Related Are We to Bananas? Pfizer. https://www.pfizer.com/news/articles/how_genetically_related_are_we_to_bananas.
Perogamvros, L., & Schwartz, S. (2012). The roles of the reward system in sleep and dreaming.
Neuroscience & Biobehavioral Reviews. https://doi.org/10.1016/j.neubiorev.2012.05.010
Siclari, F., Baird, B., Perogamvros, L., Bernardi, G., Larocque, J. J., Riedner, B., . . . Tononi, G. (2017). The neural correlates of dreaming. Nature Neuroscience. https://doi.org/10.1038/nn.4545
Xu, X., Yang, W., Tian, B., Sui, X., Chi, W., Rao, Y., & Tang, C. (2021). Quantitative investigation reveals distinct phases in Drosophila sleep. Communications Biology, 4(1), 364. https://doi.org/10.1038/s42003-021-01883-y
Yap, M., Grabowska, M. J., Rohrscheib, C., Jeans, R., Troup, M., Paulk, A. C., Bart van Alphen,
Shaw, P. J., & Bruno van Swinderen. (2017). Oscillatory brain activity in spontaneous
and induced sleep stages in flies. Nature Communications.
https://doi.org/10.1038/s41467-017-02024-y
